Lipid Matters - A Personal Blog
Or "Lipids Matter". An occasional series of notes on publications or other items dealing with lipid science that seem to be of particular interest to the originator of this web page, Bill Christie. Inevitably, the selection is highly personal and subjective. The older entries are archived for at least a year in a separate web page here..
August 30th, 2017
 Every week there is a report in the literature of a novel lipid being found in some exotic organism.
Perhaps more surprising is how often new lipid structures are revealed in human tissues, and there are two good examples this week.
While improvements in technology are often behind new discoveries, another explanation is that the authors have simply looked closer at minor components,
or perhaps it is a bit of both. Ion mobility mass spectrometry appears to offer new opportunities in terms of separation
and analysis of complex glycosphingolipids according to the charge state, the carbohydrate chain length and the degree of sialylation or other substitution
with no requirement for a chromatography step, and a new report describes an application to brain lipids
in which a large numbers of novel gangliosides modified with acetyl groups were discovered (Sarbu, M. et al. Electrospray
ionization ion mobility mass spectrometry provides novel insights into the pattern and activity of fetal hippocampus gangliosides.
Biochimie, 139, 81-94 (2017); DOI).
Last year, I commented on a paper from the same laboratory, where novel sialylated gangliosides were found in fetal brain by the same methodology.
Every week there is a report in the literature of a novel lipid being found in some exotic organism.
Perhaps more surprising is how often new lipid structures are revealed in human tissues, and there are two good examples this week.
While improvements in technology are often behind new discoveries, another explanation is that the authors have simply looked closer at minor components,
or perhaps it is a bit of both. Ion mobility mass spectrometry appears to offer new opportunities in terms of separation
and analysis of complex glycosphingolipids according to the charge state, the carbohydrate chain length and the degree of sialylation or other substitution
with no requirement for a chromatography step, and a new report describes an application to brain lipids
in which a large numbers of novel gangliosides modified with acetyl groups were discovered (Sarbu, M. et al. Electrospray
ionization ion mobility mass spectrometry provides novel insights into the pattern and activity of fetal hippocampus gangliosides.
Biochimie, 139, 81-94 (2017); DOI).
Last year, I commented on a paper from the same laboratory, where novel sialylated gangliosides were found in fetal brain by the same methodology.
The second relevant report is of the discovery of novel cholesterol esters containing estolide bound fatty acids in vernix caseosa, the natural biofilm on the skin of new-born babies (Kalužíková, A. et al. Cholesteryl esters of ω-(O-acyl)-hydroxy fatty acids in vernix caseosa. J. Lipid Res., 58, 1579-1590 (2017); DOI). By means of reversed-phase liquid chromatography linked to mass spectrometry with atmospheric pressure chemical ionization, approximately 300 molecular species of this new lipid class were identified, with the most abundant containing a 32:1 ω-hydroxy fatty acid linked to those of a more conventional kind. I was aware of vernix caseosa as a source of wax esters containing a complex mixture of branched-chain fatty acids, but they are are very different in nature from these new lipids.
August 16th, 2017
Phosphatidylcholine and phosphatidylethanolamine can hardly be considered as neglected as they are the most abundant lipids in most cellular membranes in animals. On the other hand, I am not sure that the full range of their biological properties other than as membrane building blocks is always recognized. A new review is certainly helpful and has enabled me to update my pages here (van der Veen, J.N. et al. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim. Biophys. Acta, Biomembranes, 1859, 1558-1572 (2017); DOI). For example, the role of phosphatidylcholine in lipoprotein metabolism is well known, but I was not aware that phosphatidylethanolamine is present in relatively high concentrations in newly secreted VLDL particles and that this lipid is almost certainly involved in VLDL assembly and/or secretion. On the other hand, it is rapidly and efficiently removed from the VLDL in the circulation but where and how?
One of my pet hates to which I refer here from time to time is the excessive and often unnecessary use of abbreviations and acronyms in scientific papers and especially when they are used in titles. They are a convenience for authors but a nuisance for readers. It seems that every technique, every enzyme, every gene and every metabolite now has its own abbreviation. Of course, I am not arguing that these be shunned entirely, and Lipid Maps, for example, have set out a set of recommended abbreviations for lipid classes that I have used from time to time and find useful especially in figures. In the publication cited above, PC and PE are used throughout in a sensible way. On the other hand, I often find an abbreviation is defined on page 2 of a paper and is then not used again until page 10 when I have to scramble back through to find what it means. A few weeks ago I mentioned that the abbreviation MGDG was used unnecessarily in the title of a publication to replace the one-word lipid class. Of course, it all depends on context; FA means 'fatty acid' in the lipid literature, but it can also mean 'Football Association' and something rather rude. PC can mean 'phosphatidylcholine', 'politically correct', 'police constable' or 'personal computer'. I am not one of the texting generation, who have their own set of abbreviations and may consider my comments are OTT ('over the top') or even ATP ('ATypical Pedantry' - I just made that up - OK). For the moment, I'll let the thought RIP.
August 9th, 2017
In writing this blog, I often allude to those lipids that are most often cited and are therefore actively researched. However, there are a few lipids that appear to be neglected in my opinion. For example, the non-acidic glycosyldiacylglycerols of animal tissues are rarely mentioned in the literature these days. There was a flurry of activity in the 1990s but little since. As they tend to be minor components, they are easily missed, although those in saliva and related secretions may have important functions in these tissues. Another explanation for the neglect may be that they are removed from lipid extracts as part of a procedure for removing glycerolipids to produce pure sphingoglycolipid preparations for analysis. While the acidic glycosyldiacylglycerol seminolipid does feature in many publications in relation to its function in male reproductive tissues, you will struggle to find much on its occurrence and function in brain and nervous tissues. In brain, some of these lipids may be produced adventitiously by the same enzymes that produce comparable sphingolipids, but they may still have distinct functions of their own.
Cytidine diphosphate diacylglycerol (CDP-DAG) is a key intermediate in the biosynthesis of phospholipids so features in innumerable biochemical studies, but what about its natural occurrence and composition in tissues. Just try to find data! Is the explanation is that its natural occurrence is too low and modern mass spectrometric methods are not sufficiently sensitive, or that it is too unstable, or that analysts are simply not looking for it?
Two important journal issues have just come to my attention - Biochimica et Biophysica Acta (BBA), Biomembranes (Volume 1859, Issue 9, Part B, Pages 1493-1748, September 2017) dealing with the topic of "Membrane Lipid Therapy: Drugs Targeting Biomembranes" and edited by Pablo V. Escribá - and Free Radical Biology and Medicine (Volume 111, Pages 1-344, October 2017) on the theme of "4-Hydroxynonenal and Related Lipid Peroxidation Products" and edited by Giuseppe Poli and Neven Zarkovic.
August 2nd, 2017
The opening sentence of a new open-access publication is thought provoking - "The major light-harvesting complex (LHCII) found in the chloroplasts of green plants contains more than half of the chlorophylls (Chl) and is the most abundant membrane protein on earth" (Seiwert, D. et al. The non-bilayer lipid MGDG stabilizes the major light-harvesting complex (LHCII) against unfolding. Sci. Rep., 7, 5158 (2017); DOI). It must also be the most important protein for advanced life on earth as all the oxygen in the atmosphere is produced as a byproduct of the photosynthesis reaction. Monogalactosyldiacylglycerols for those of you unfamiliar with the abbreviation in the title have a conical structure and do not form bilayers, but their shape appears to match that of the trimeric LHCII complex and stabilize it while modulating the folding, conformation and function of the protein components.
The importance of the physical properties of lipids to the functions of animal organs is also illustrated by two publications in a recent issue of the Journal of Biological Chemistry. Over the years, I have read innumerable suggestions as to why docosahexaenoic acid (DHA) is important in tissues, but especially in relation to visual acuity. It now appears that its role in phospholipids is primarily to maintain the disc shape in photoreceptor cells (Shindou, H. et al. Docosahexaenoic acid preserves visual function by maintaining correct disc morphology in retinal photoreceptor cells. J. Biol. Chem., 292, 12054-12064 (2017); DOI). Cellular membrane containing DHA in the phospholipids are more flexible than those containing arachidonic acid and other fatty acids, and they may also increase the stability and function of rhodopsin. Similarly, during spermatogenesis, DHA-containing phospholipids provide membranes in spermatids with the physicochemical properties needed for normal cellular processes (Iizuka-Hishikawa, Y. et al. Lysophosphatidic acid acyltransferase 3 tunes the membrane status of germ cells by incorporating docosahexaenoic acid during spermatogenesis. J. Biol. Chem., 292, 12065-12076 (2017); DOI). The second of these papers is the authors' choice and therefore open access.
The latest issue of the journal Molecular Aspects of Medicine (Volume 56, Pages 1-110 (August, 2017)) deals with the theme of "Bile acids, roles in integrative physiology and pathophysiology" (edited by David H. Volle).
July 26th, 2017
 Following
on from last week's blog, a paper on protein S-palmitoylation has caught my attention.
New methodology involving a site-specific acyl-biotin-exchange reaction for the complete palmitoylated-proteome of a tissue
has enabled the identification of what appears to me at least to be an extraordinary number of palmitoylation sites in brain tissue
(Collins, M.O. et al. Global, site-specific analysis of neuronal protein S-acylation.
Sci. Rep., 7, 4683 (2017); DOI).
490 Palmitoylation sites have been identified on 342 synaptic proteins, 44% of which are integral membrane proteins.
It is now apparent that protein palmitoylation is essential for intracellular signalling and for the folding,
trafficking and function of such disparate proteins as Src-family kinases, Ras family GTPases, G-proteins and G-protein coupled receptors.
Many of the palmitoylation sites co-located with phosphorylation sites, and it seems to me that the biochemical world must now regard
protein palmitoylation-depalmitoylation in the same light as phosphorylation-dephosphorylation in the regulation of enzyme activity.
Following
on from last week's blog, a paper on protein S-palmitoylation has caught my attention.
New methodology involving a site-specific acyl-biotin-exchange reaction for the complete palmitoylated-proteome of a tissue
has enabled the identification of what appears to me at least to be an extraordinary number of palmitoylation sites in brain tissue
(Collins, M.O. et al. Global, site-specific analysis of neuronal protein S-acylation.
Sci. Rep., 7, 4683 (2017); DOI).
490 Palmitoylation sites have been identified on 342 synaptic proteins, 44% of which are integral membrane proteins.
It is now apparent that protein palmitoylation is essential for intracellular signalling and for the folding,
trafficking and function of such disparate proteins as Src-family kinases, Ras family GTPases, G-proteins and G-protein coupled receptors.
Many of the palmitoylation sites co-located with phosphorylation sites, and it seems to me that the biochemical world must now regard
protein palmitoylation-depalmitoylation in the same light as phosphorylation-dephosphorylation in the regulation of enzyme activity.
My enthusiasm for the potential of bacterial lipopeptides as a source of new antibiotics (see last week also) has taken something of a blow with a new publication describing the practical difficulties in recovering them from natural sources (Coutte, F. et al. Microbial lipopeptide production and purification bioprocesses, current progress and future challenges. Biotechn. J., 12, 1600566 (2017); DOI). There are three major challenges: bacteria produce quorum-sensing molecules that sense cell density and thence limit their growth - the more important of these are in fact lipids, i.e. N-acylhomoserine lactones. Secondly there are problems of foam production because of the amphiphilic nature of the products that cause handling difficulties, and finally the complex mixtures formed are not easily resolved into single components. It may take time but I suspect these problems will eventually be overcome.
Both publications cited this week are open access. Incidentally, I maintain a rough log of my updates to my Lipid essentials pages here. Last year sphingosine 1-phosphate and phosphoinositides received most attention, this year so far it is proteolipids and isoprostanes.
July 19th, 2017
In the search for new antibiotics, lipopeptides appear to offer great potential if problems of toxicity can be overcome. Paenibacillus sp. have proved to be of special interest, and a new report describes a fresh isolate that produces novel cyclic and linear lipopeptides, both of which have antibiotic activity against Gram-negative and Gram-positive bacteria (Huang, E. et al. New Paenibacillus strain produces a family of linear and cyclic antimicrobial lipopeptides: cyclization is not essential for their antimicrobial activity. FEMS Microbiol. Letts, 364, fnx049 (2017); DOI). Much of the emphasis of recent work has been on cyclic lipopeptides, but chemical synthesis of linear lipopeptides is much easier technically than of cyclic equivalents so this should open up opportunities for the design and testing of new families of related molecules for their therapeutic value.
When I was revising my web page on protein acylation (proteolipids) recently, I became aware that I had written much less on N-myristoylation than on S-palmitoylation, and this was reflected in the reading list at the end. On thinking it over, I believe this is because the latter is a more dynamic modification, the regulation of which can be seen to be relevant to a host of metabolic processes. Indeed, one element of the regulation of the activity of N-myristoylated proteins is additional S-palmitoylation/deacylation reactions. I was able to redress the balance a little after reading a new open access publication (Udenwobele, D.I. et al. Myristoylation: an important protein modification in the immune response. Front. Immunol., 8, 751 (2017); DOI). Incidentally, a second open access review in this general area was published this week (Chen, J.J. and Boehning, D. Protein lipidation as a regulator of apoptotic calcium release: relevance to cancer. Front. Oncol., 7, 138 (2017); DOI).
July 12th, 2017
It is astonishing how the view of lipids held by biochemists has changed in the last 50 years. I have to confess that I did not always recognize each milestone in lipid science as it was achieved but I can look back now in admiration of the work of so many of my contemporaries. One such is William Dowhan who has just described his career and research philosophy in an open access publication (Dowhan, W. Understanding phospholipid function: Why are there so many lipids? J. Biol. Chem., 292, 10755-10766 (2017); DOI). While signalling was a major focus for research in the lipid field over the period, Dowhan was instead pioneering the study of how lipids interact with proteins to modify their functions using E. coli as his model organism to reveal "direct lipid-protein interactions that govern dynamic structural and functional properties of membrane proteins". I can recommend this as a good read both for the science and as a personal record of a distinguished career. Incidentally, he published a review with a very similar title back in 1997, and it is fascinating to learn what has been accomplished since then.
I was not around when cholesterol was discovered and a new open access review marks the 200th anniversary of the recognition by the great French chemist Michel Chevreul that it was a non-saponifiable lipid present in gall stones (Chaudhuri, A. and Anand, D. Cholesterol: Revisiting its fluorescent journey on 200th anniversary of Chevruel's "cholesterine". Biomed. Spectr. Imaging, 6, 1-24 (2017); DOI). Aside from the fascinating historical introduction (in which the subject's name is unfortunately misspelt), this open access publication describes the use of fluorescent probes in studying cholesterol function in cells. My former mentor Frank Gunstone kept a picture in his office of Chevreul at work in his laboratory in his 100th year, and I reproduce it below. Now there is an ambition!

If I am to achieve this, it seems that I have to keep up my fish and presumably fish oil consumption (Zeng, L.F. et al. An exploration of the role of a fish-oriented diet in cognitive decline: a systematic review of the literature. Oncotarget, 8, 39877-39895 (2017); DOI).
July 5th, 2017
Eric Murphy makes a cogent plea for respect for copyright in an editorial in the latest issue of Lipids (Murphy, E.J. An ethical dilemma: to share or not to share your paper published in Lipids using an on-line outlet. Lipids, 52, 573-574 (2017); DOI). Posting papers to sites such as ResearchGate is a breach of copyright if the paper is not already open access and is undoubtedly illegal. He suggests that rather than doing this authors should use open access journals if they feel strongly about freedom of use. While I am sympathetic to much of what he says, I do not believe that the problem can be discussed entirely in such black and white terms. If I email an author and ask for a pdf file of a paper in the same way as years ago I might have requested a reprint, this probably comes into the category of fair use, but if we extend this to consider a correspondent who sends me a pdf file of a paper not his own to which I do not have immediate access, should I have to search my conscience? Who am I cheating; there is no way that as a private retiree I could consider spending up to £40 as demanded by publishers for a pdf file that may or may not be of use to me (though if it were £2 I might). If I accept an 'illegal' copy, I will not distribute it elsewhere and I will cite it in this website so the authors and the journal get some publicity at least.
Ethics aside, it is hard to feel sorry for scientific publishers, some of whom are apparently making huge profits on turnover according to an article in the Guardian newspaper. For example in 2010, Elsevier made a 36% profit on turnover. When you consider that they do not have to pay anything to authors or referees this seems grossly excessive. On the other hand, I have no sympathy for sites such as Sci-Hub, who according to Nature News have just been ordered by a US court to pay US$15 million in damages to Elsevier for copyright infringement, although the latter are unlikely to see any of this money as the site is run out of the jurisdiction of the court in Russia. As I understand it, this site is still operating and largely offering preprints of papers without charge, although they are aggressive in seeking donations (assuming anyone is willing to send bank/credit card details to Russia). Incidentally, the problem is not new in that I recently read a biography of Charles Dickens, who was greatly aggrieved because US publishers reprinted his books as soon as they could get their hands on them without paying him royalties.
What is the answer? Apart from having more open access journals and papers, I would be content if more publishers allowed access to back content after 1-2 years as is already the case with many non-commercial journals especially those with a biological remit. It seems wrong that I am not able to have digital copies of my own papers in journals published by the Royal Society of Chemistry in the 1960s without paying a hefty fee.
June 28th, 2017
 The
journal Biochim. Biophys. Acta - Molecular and Cell Biology of Lipids has a special Issue for August
just online entitled "BBALIP_Lipidomics Opinion Articles" and edited by Sepp Kohlwein.
At first glance, there seems to be a wide and diverse range of topics going beyond the technical aspects into the biology.
While I am fascinated and a little envious of the new methodology, I am more interested in the results.
So far, I have only had time to look at one of the reviews, which is highly relevant to my Lipid Essentials pages here
in relation to presentation of data (Liebisch, G. et al. Reporting of lipidomics data should be standardized.
Biochim. Biophys. Acta, 1862, 747-751 (2017); DOI).
Many of the points made seem sensible, including the suggestion that data should be reported in terms of absolute amounts
although I am not clear whether they mean by weight or in terms of molar amounts.
They also suggest that data should be available in spreadsheets rather than Word documents or pdf files to make inter-laboratory comparisons easier.
The
journal Biochim. Biophys. Acta - Molecular and Cell Biology of Lipids has a special Issue for August
just online entitled "BBALIP_Lipidomics Opinion Articles" and edited by Sepp Kohlwein.
At first glance, there seems to be a wide and diverse range of topics going beyond the technical aspects into the biology.
While I am fascinated and a little envious of the new methodology, I am more interested in the results.
So far, I have only had time to look at one of the reviews, which is highly relevant to my Lipid Essentials pages here
in relation to presentation of data (Liebisch, G. et al. Reporting of lipidomics data should be standardized.
Biochim. Biophys. Acta, 1862, 747-751 (2017); DOI).
Many of the points made seem sensible, including the suggestion that data should be reported in terms of absolute amounts
although I am not clear whether they mean by weight or in terms of molar amounts.
They also suggest that data should be available in spreadsheets rather than Word documents or pdf files to make inter-laboratory comparisons easier.
In my articles here, I do not quote any analytical data made by modern mass spectrometric methods - all come from papers published in the 70s and 80s when the methodology was more time consuming but capable of high precision. The problem is that data obtained now in terms of molecular species compositions are in a format that does not lend itself to simple presentation. A phospholipid with 10 fatty acids can exist in the form of 90 molecular species, including positional isomers on the glycerol moiety, while a similar triacylglycerol can have 500 species not including enantiomers. When I came into lipid science, we were more concerned with positional distributions of fatty acids as determined following hydrolytic cleavage with specific lipases; analysis of molecular species was often secondary. It was a simple task to tabulate data for the fatty acid composition of each position in a phospholipid as two columns of fatty acids normalized to 100 mol% with roughly 10 numbers in each. Comparison of data from other laboratories was straight forward, and if you need examples look at almost any of the tables in my web page here on triacylglycerol compositions where data from several sources are presented in a single table.
Such positional data are relevant to biosynthetic processes, hydrolysis by enzymes and lipid remodelling. To give just two examples, arachidonic acid from position sn-2 of phospholipids is used for eicosanoid production while that from position sn-1 is used for anandamide biosynthesis. Of course, molecular species data are important also but this may not be as immediately obvious.
Although mass spectrometric methodology produces data in the form of amounts of the various molecular species, is it necessarily to present it in this form only? Lipidomics methodology is available to determine positional distributions of fatty acids on the glycerol moiety in each species, and while I am not up to date on the mechanics of this there are certainly plenty of papers on the topic. I suspect that the older methods may be capable of greater precision, but mass spectrometry may be good enough for comparative purposes. If so, would it not be possible to add simple mathematical formulae to the spreadsheets to generate tables of positional data for the fatty acids in each lipid class from the molecular species data? Data in both formats are important, but a simple comparison of positional data for each lipid as a first step in interpretation might point to the areas of the molecular species information that require a closer examination. It would certainly simplify interlaboratory comparisons.
June 21st, 2017
In this blog, I have often discussed the therapeutic potential of particular lipids against human diseases. It may be worth a reminder that lipids can have similar beneficial functions in plants. For example, plants in the Solanacea and other families have glandular trichomes, i.e. secretory organs on the external surfaces that secrete mixtures of sugar esters onto the plant aerial surfaces that act as protective agents against both insect herbivores and pathogenic fungi Luu, V.T. et al. O-Acyl sugars protect a wild tobacco from both native fungal pathogens and a specialist herbivore. Plant Physiol., 174, 370-386 (2017); DOI). In the tomato, for example, these metabolites consist of a carbohydrate backbone, usually glucose or sucrose, to which two to five fatty acids are esterified. The aliphatic acyl chains vary in length from C2 to C12 and are straight chain or have iso- or anteiso-methyl-branches.
My open access publication of the week is perhaps more mainstream and deals with the role of sphingolipids in brain development (Olsen, A.S.B. and Færgeman, N.J. Sphingolipids: membrane microdomains in brain development, function and neurological diseases. Open Biol., 7, 170069 (2017); DOI).
In the discussion of the new biologically active lipids 'FAHFA' (Fatty Acid Hydroxy Fatty Acid), I do not recall the term "estolide" mentioned although this has been in use since at least the 1950s (according to Google scholar). The definition from the review cited here is "they are intermolecular esters comprised of at least two fatty acid molecules". In animals, the best known example is skin ceramides, but they are also present in bacteria (ornithine lipids and lipid A), many seed oils and yeast. However, the FAHFA are distinctive and differ from the rest in that they have a free carboxyl group. As an example, it may seem something of a misnomer, but hexaacyl triacylglycerols were reported from ergot oil, i.e. with three moles of ricinoleate attached to glycerol each of which is esterified with a long chain fatty acid (Morris, L.J. and Hall, S.W. Structure of glycerides of ergot oils. Lipids, 1, 188-196 (1966); DOI). In fact, estolides are important industrial products with applications in lubricants (Zerkowski, J.A. Estolides: From structure and function to structured and functionalized. Lipid Technology, 20, 253-256 (2008); DOI). Incidentally, this review suggests that the first description of an estolide may have been "a 1915 report in Die Naturwissenschaften mentioning their isolation from conifer needles".
I was not around in 1915, but I do remember Lindsay Morris - the author of the 1966 paper. He was a few years ahead of me first as a PhD student with Frank Gunstone and then as a post doc with Ralph Holman, so I knew him first simply as a legendary figure for his activities both within and out of the lab. He will be best remembered as one of the inventors of silver ion chromatography. When we did eventually meet, I found him an engaging person with boundless energy and enthusiasm. I understand that he moved back to Scotland when he retired from Unilever Research, and sadly he died a few years ago.
June 14th, 2017
In my essays here, I have used a rather strict definition of what constitutes an endocannabinoid, i.e. that they must interact with the cannabinoid receptors CB1 and CB2. Thus of the amides, anandamide is obviously an endocannabinoid as is oleamide, but oleoylethanolamide is not. For many purposes this is a useful practical distinction, but there are grey areas and I wonder if I have been too pedantic especially as the 'true' endocannabinoids interact with a number of other receptors. Perhaps we need a new collective term that embraces all the fatty acid amides and simple lipaminoacids - 'amidolipins'? For example, of the other amides palmitoylethanolamide does not interact with the CB1 and CB2 receptors to a significant extent, but it has does have synergistic or "entourage" effects with the 'true' endocannabinoids. This interesting lipid exerts many biological effects in its own right, apparently by a multiplicity of mechanisms and receptors that impinge upon the activities of the other acyl amides. It is undergoing clinical trials for the relief of chronic pain and is the subject of a new review (Petrosino, S. and Di Marzo, V. The pharmacology of palmitoylethanolamide and first data on the therapeutic efficacy of some of its new formulations. Brit. J. Pharmacol., 174, 1349-1365 (2017); DOI).
The N-acylserotonins are another class of fatty amides that also fall into a grey classification area, simply because we do not yet appear to know with which receptors they interact. In particular, it is now clear that N-docosahexaenoylserotonin is present in human intestinal tissue and is a potent anti-inflammatory mediator that may be relevant to intestinal inflammatory conditions such as Crohn's disease and ulcerative colitis. It is a fascinating addition to the list of lipids containing polyunsaturated fatty acids of the (n-3) family with beneficial properties (Wang, Y. et al. Docosahexaenoyl serotonin emerges as most potent inhibitor of IL-17 and CCL-20 released by blood mononuclear cells from a series of N-acyl serotonins identified in human intestinal tissue. Biochim. Biophys. Acta, 1862, 823-831 (2017); DOI).
In relation to the 'true' endocannabinoids, a new review suggests that some of their biological properties may be mediated through the production of nitric oxide, which functions as a versatile signalling intermediate and is ubiquitous in tissues (Lipina, C. and Hundal, H.S. The endocannabinoid system: ‘NO’ longer anonymous in the control of nitrergic signalling? J. Mol. Cell Biol. 9, 91-103 (2017); DOI).
June 7th, 2017
The presence of α-galactosylceramide as opposed to the β-form in human tissues and its astonishing biological activity as an anti-tumor immunotherapeutic agent has been one of the pleasant surprises of this year (and has featured earlier this year in this blog). Indeed, I understand that it is undergoing clinical trials as an anti-tumor agent. One major difficulty in studying its metabolism and function is the low levels at which it occurs naturally in tissues (0.02% of the total galactosylceramides in RBL-CD1d cells, for example). A new LC-MS2 separation of the stereoisomers has now been described that appears to solve the problem (von Gerichten, J. et al. Diastereomer-specific quantification of bioactive hexosylceramides from bacteria and mammals. J. Lipid Res., 58, 1247-1258 (2017); DOI). As this lipid is produced by intestinal bacteria, it is a useful reminder after my previous two blogs that bacteria have many virtues and they are not always harmful. It is a truism that advances in methodology often lead to advances in the science, so watch this space. If I want to be picky, I would raise my old chestnut that the term "hydrophilic interaction chromatography" applied to the separation is meaningless unless we know more about the nature of the stationary phase. In fairness to authors, the manufacturers are often silent on this point.
The Journal of Steroid Biochemistry and Molecular Biology has published a special issue on the topic of "Oxysterols: Players in Different Metabolic Leagues" (Volume 169, Pages 1-234 (May 2017)) and edited by Luigi Iuliano, Dieter Lütjohann, Gérard Lizard and Ingemar Bjorkhem.
May 31st, 2017
 We can never fully free ourselves of the national and occupational stereotypes that are part of our cultural heritage,
and I am sure my readers will have an idea at the back of their mind of a "typical" Scotsman.
Our view of Russians and scientists is of a very serious and possibly humorless people,
so it was a rather pleasant surprise to find an item in Nature News
( DOI ) regarding a 'Monument to an Anonymous Peer Reviewer' outside
the Higher School of Economics in Moscow.
Immortalized in concrete, "the sculpture takes the form of a die displaying on its five visible sides the possible results of review
- 'Accept', 'Minor Changes', 'Major Changes', 'Revise and Resubmit' and 'Reject'."
One more stereotype bites the dust.
We can never fully free ourselves of the national and occupational stereotypes that are part of our cultural heritage,
and I am sure my readers will have an idea at the back of their mind of a "typical" Scotsman.
Our view of Russians and scientists is of a very serious and possibly humorless people,
so it was a rather pleasant surprise to find an item in Nature News
( DOI ) regarding a 'Monument to an Anonymous Peer Reviewer' outside
the Higher School of Economics in Moscow.
Immortalized in concrete, "the sculpture takes the form of a die displaying on its five visible sides the possible results of review
- 'Accept', 'Minor Changes', 'Major Changes', 'Revise and Resubmit' and 'Reject'."
One more stereotype bites the dust.
Phosphatidylserine is known to have an important role in the regulation of apoptosis or programmed cell death, the natural process by which aged or damaged cells are removed from tissues before they can exert harmful effects. A new review (open access) gives a clear explanation of the process in relation to erythrocytes, where phosphatidylserine is located in the inner leaflet of the membrane bilayer under low Ca2+ conditions when a phospholipid scramblase is suppressed by membrane cholesterol, but it is exposed to the outer leaflet under elevated Ca2+ concentrations which activate the scramblase (Arashiki, N. and Takakuwa, Y. Maintenance and regulation of asymmetric phospholipid distribution in human erythrocyte membranes: implications for erythrocyte functions. Curr. Opinion Hemat., 24, 167-172 (2017); DOI). The phosphatidylserine on the outer leaflet of the cell is then recognized by a receptor on the surface of macrophages and related scavenger cells, and these proceed to remove the apoptotic cells in a non-inflammatory manner.
Last week, I discussed two reviews that dealt with the sneaky ways pathogens made use of the lipids of their hosts for their own nefarious purposes. A new review that has the virtue of being open access discusses this in relation to cholesterol specifically (Samanta, D. et al. Manipulation of host cholesterol by obligate intracellular bacteria. Front. Cell. Inf. Microbiol., 7, 165 (2017); DOI). To gain entry into cells, pathogens utilize the cholesterol-rich microdomains in membranes known as rafts. Then, it is apparent that they can manipulate host cholesterol metabolism, including uptake, efflux, and storage, to access nutrient-rich vesicles or acquire membrane components. They also hijack the host cell signaling pathways involving cholesterol that are favorable for their intracellular survival.
The Journal of Experimental Botany has a special issue devoted to the "The Flowering of Jasmonate Research" (1 March, 2017).
May 24th, 2017
A week in the Canary Islands has taken my mind off lipid science for a time, but now I have twice as many papers as usual to read. A catchy title to a review often heralds a more entertaining discussion to follow as in this instance (Pathak, D. and Mallik, R. Lipid - motor interactions: soap opera or symphony? Curr. Opinion Cell Biol., 44, 79-85 (2017); DOI). Motor proteins are here defined as "ATPases that convert chemical energy into mechanical energy to drive many cellular functions including intracellular transport of vesicles". These enzymes require interactions with specific lipids, especially the phosphoinositides and cholesterol, to direct them to specific membranes. As to "soap opera etc", read the first paragraph for an elegant explanation of the analogy. The protein can have highly specific binding sites for particular lipids or it can indirectly react to membrane curvature induced by characteristic lipid head groups. Here much of the discussion focuses on the endosome/phagosome compartment partly because of the importance to normal cellular metabolism, and partly because pathogens in phagosomes use the lipid interactions to survive in host cells. Incidentally, there is a rather substantial new (if relatively inaccessible) book chapter that relates to the latter process (Fozo, E.M. and Rucks, E.A. The making and taking of lipids: the role of bacterial lipid synthesis and the harnessing of host lipids in bacterial pathogenesis. Adv. Microb. Physiol., 69, 51-155 (2016); DOI).
I am a spectator only to modern mass spectrometric techniques, but I have the impression that advances in software and data management are as important as those in instrumentation in driving what can now be achieved. For example, LipidFinder optimizes analysis based on users' own data, and a new open access publication describes its use to identify three 12-hydroxyeicosatetraenoic acid phosphoinositides in thrombin-activated platelets (O'Connor, A. et al. LipidFinder: A computational workflow for discovery of lipids identifies eicosanoid-phosphoinositides in platelets. JCI Insight, 2, e91634 (2017); DOI). Another research group describes the use of the software IE-Omics to automate data acquisition by MS/MS in sequential injections to improve the coverage of the lipidome especially with regard to trace species (Koelmel, J.P. et al. Expanding lipidome coverage using LC-MS/MS data-dependent acquisition with automated exclusion list generation. J. Am. Soc. Mass Spectrom., 28, 908-917 (2017); DOI).
The therapeutic potential of docosanoids such as the resolvins is increasingly becoming evident, and an application of 17(R)-hydroxy-docosahexaenoic acid to the relief of pain in osteoarthritis, if not yet the underlying cause, in animal models is described in a new publication hopefully as a prelude to clinical studies (Huang, J.T. et al. Targeting the D series resolvin receptor system for the treatment of osteoarthritis pain. Arthritis Rheumatol., 69, 996-1008 (2017); DOI).
May 10th, 2017
There is a short series of reviews on the theme of 'Lipid Methodology' (edited by Howard Goldfine and Ziqiang Guan) in a recent issue of Analytical Biochemistry (Volume 524, Pages 1-76 (1 May 2017)). One of these dealing with cholesterolomics is open access, but the one that caught my eye especially deals with the various modes of high-performance liquid chromatography that can be applied for the separation of regio- and stereoisomers of triacylglycerols (Rezanka et al. Regioisomeric and enantiomeric analysis of triacylglycerols. Anal. Biochem., 524, 3-12 (2017); DOI). Modern mass spectrometric methods dominate the recent analytical literature, but they cannot accomplish stereospecific analysis of triacyl-sn-glycerols, so I am always encouraged to see that alternative methods are still being pursued.
Some months ago, my attention was drawn to the fact that some subjects were being reviewed to exhaustion, with more than 20 reviews a year devoted to each of the topics of phosphoinositides and sphingosine-1-phosphate in particular. Of course, the reason these topics receive so much attention is because they are so dynamic and there is much important new research to discuss. Therefore, I make no apology for drawing your attention to a new review dealing with sphingosine-1-phosphate, which among its many virtues is open access (Pyne, N.J. and Pyne, S. Sphingosine 1-phosphate receptor 1 signaling in mammalian cells. Molecules, 22, 344 (2017); (2017); DOI).
May 3rd, 2017
One of the more surprising lipid discoveries in recent years has been fatty acids with a centrally located hydroxyl group to which a further fatty acid is linked as an estolide or 'FAHFA' (Fatty Acid Hydroxy Fatty Acid), such as the palmitoyl ester of 9-hydroxy-stearic acid (note that both component fatty acids are fully saturated), which was first found in the adipose tissue of mice. These have anti-diabetic and anti-inflammatory effects, even when administered orally, and they protect against colitis by regulating gut innate and adaptive immune responses. Although details of the biosynthesis have still to be established, there seems little doubt that they are formed endogenously as a new study has established that the hydroxyl group has defined stereochemistry, i.e. it is of the R-configuration (Nelson, A.T. et al. Stereochemistry of endogenous palmitic acid ester of 9-hydroxystearic acid and relevance of absolute configuration to regulation. J. Am. Chem. Soc., 139, 4943-4947 (2017); DOI). Unfortunately, I am dependent on the abstract for this information as access is closed to non-subscribers.
I try to keep up with with the plethora of new eicosanoids and docosanoids that continue to be discovered, but I did not realize that more than 70 oxygenated metabolites of docosahexaenoic acid (DHA) had been discovered to date as summarized in a new review (Kuda, O. Bioactive metabolites of docosahexaenoic acid. Biochimie, 136, 12-20 (2017); DOI). Included among these are FAHFA derived from essential fatty acids, i.e. with 14-hydroxydocosahexaenoic acid (14-OH-DHA) esterified to 9- and 13-hydroxyoctadecadienoic acids, for example; these have profound anti-inflammatory effects.
A few weeks ago, I pointed out here that the Journal of Lipid Research had taken a retrograde step in closing access to papers that had been accepted but were still in manuscript form. Either this was a technical error or they have seen the error of their ways and this policy has now been reversed.
April 26th, 2017
 Urine samples are usually regarded as the easiest non-invasive method of obtaining samples for analysis,
but then any metabolites have passed through the kidney and may have been substantially altered.
I had not considered human tears for this purpose, but it seems that tear fluid can serve as a means to identify and monitor novel biomarkers
in ocular and systemic disease, and in particular the specialized pro-resolving mediators (SPMs).
For example, resolvin D1, protectin D1, lipoxin A4, and resolvin E1 are accessible in this way in quantities that are known to
be active biologically (English, J.T. et al. Identification and profiling of specialized
pro-resolving mediators in human tears by lipid mediator metabolomics. PLEFA, 117, 17-27 (2017);
DOI).
When I first saw the title, it brought to mind the old Julie London song "Cry me a River", but it seems that only 100μl of tears
obtained "through an induction of an emotional response" were required for the identification and quantification of 21 different metabolites.
This seems an astonishing example of the sensitivity of modern mass spectrometric methodology.
The results are also surprising in that while SPMs were detected in male donors, they were essentially absent in females.
Incidentally, if any of my younger readers don't know the above song (or the Barbra Streisand version), a treat awaits you.
Urine samples are usually regarded as the easiest non-invasive method of obtaining samples for analysis,
but then any metabolites have passed through the kidney and may have been substantially altered.
I had not considered human tears for this purpose, but it seems that tear fluid can serve as a means to identify and monitor novel biomarkers
in ocular and systemic disease, and in particular the specialized pro-resolving mediators (SPMs).
For example, resolvin D1, protectin D1, lipoxin A4, and resolvin E1 are accessible in this way in quantities that are known to
be active biologically (English, J.T. et al. Identification and profiling of specialized
pro-resolving mediators in human tears by lipid mediator metabolomics. PLEFA, 117, 17-27 (2017);
DOI).
When I first saw the title, it brought to mind the old Julie London song "Cry me a River", but it seems that only 100μl of tears
obtained "through an induction of an emotional response" were required for the identification and quantification of 21 different metabolites.
This seems an astonishing example of the sensitivity of modern mass spectrometric methodology.
The results are also surprising in that while SPMs were detected in male donors, they were essentially absent in females.
Incidentally, if any of my younger readers don't know the above song (or the Barbra Streisand version), a treat awaits you.
Another record appears to have been broken in that trace levels of highly unsaturated fatty acids of the (n-3) family suggested to be 38:7(n-3) to 44:12(n-3) have been reported from brains of patients with genetic impairments of peroxisome function. The last must be the most highly unsaturated fatty acid of conventional origin known (Takashima, S. et al. Detection of unusual very-long-chain fatty acid and ether lipid derivatives in the fibroblasts and plasma of patients with peroxisomal diseases using liquid chromatography-mass spectrometry. Mol. Gen. Metab., 120, 255-268 (2017); DOI).
April 19th, 2017
Some years ago, I took issue with the LipidMaps consortium over some aspects of their lipid classification system. In particular, I though they were wrong to create a distinct class for glycerophospholipids while lumping glycosyldiacylglycerols in with triacylglycerols. As glycosyldiacylglycerols can substitute for phospholipids under conditions of phosphate deprivation in plants and both function exclusively in membranes, I believed that both groups should have the same ranking. Apparently, I did not persuade them as the ranking did not change. One lipid that is particularly anomalous in this classification is the highly polar plant sulfolipid sulfoquinovosyldiacylglycerol, which cannot be compared with storage lipids in any respect; it is the subject of a new review (Goddard-Borger, E.D. and Williams, S.J. Sulfoquinovose in the biosphere: occurrence, metabolism and functions. Biochem. J., 474, 827-849 (2017); DOI). As access is closed to non-subscribers, I am grateful to a friend for giving me a sight of it. While I was familiar with much of what the authors had to say about the biochemistry and function, I was not aware that it was such an important component of the sulfur cycle in the biosphere. The total annual synthesis of sulfolipid is thought to be of the order of 1013 kg per annum!
The March issue of Biochimie (Volume 134, Pages 1-138 (March 2017)) contains a number of articles with the theme of brown fat metabolism (Edited by Frédéric Bouillaud, Louis Casteilla, Susanne Klaus and Bruno Miroux). The May issue of this journal (Volume 136, pages 1-104) is devoted to "Pleiotropic physiological roles of PPARs and fatty acids: A tribute to Paul Grimaldi" (edited by Nada A. Abumrad, Ez-Zoubir Amri, Serge Luquet and Claude Forest).
Fatty acid binding proteins (FABPs) are a family of small cytoplasmic proteins that are highly conserved and as the name suggests bind long-chain fatty acids; they facilitate the transfer of fatty acids between extra- and intracellular membranes and receptors. Of these, FABP7 is located in astrocytes of the brain and binds DHA with the highest affinity. It is now reported to be required for normal sleep in humans and other animals. There is a brief popular report in Sci News with a link to the original article.
I have come across the Latin expression "in silico" in the titles of several recent publications, and I now understand that this means "in silicon" strictly speaking but is used to mean "performed on computers or via computer simulation". Those ancient Romans were cleverer than I would have believed if they knew of silicon and anticipated the use of computers.
April 12th, 2017
An item in Nature News drew my attention to Unpaywall - a free web-browser extension that hunts for papers in more than 5,300 repositories worldwide, including preprint servers and institutional databases, to find freely accessible (and legal) copies of research articles. It is an add-on to Firefox or Google Chrome that is quick and easy to install and use. In a brief trial, it found me one paper quickly that I needed to update these pages, and just as importantly found that two of interest were not available yet as open access so saving time in fruitless searching. The article in Nature suggests that more such tools are on the way.
The titles of some publication simply shout for attention (e.g. Li, X.B. et al. The slim, the fat, and the obese: guess who lives the longest? Current Genetics, 63, 43-49 (2017); DOI). It appears that there is a phenomenon called the "obesity paradox" in that the overweight population enjoys the lowest rate of mortality from all causes in contradiction to everything that scientists and clinicians think they know. The authors believe that the answer may lie in a new cytoprotective function of triacylglycerols. As a relatively lean 140 pounder, should I be worried? Certainly, I wont feel guilty next time I am offered a big sugary doughnut with my morning coffee.
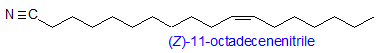
Fascinating new lipids are discovered all the time, but occasionally nature springs a nasty surprise. The latest in this category are alkyl cyanides produced by bacterial species. These can be either unbranched saturated or unsaturated with an omega-7 double bond, such as (Z)-11-octadecenenitrile, or methyl-branched unsaturated cyanides with the double bond located at C-3, such as (Z)-13-methyltetradec-3-enenitrile. Fatty acids are the biosynthetic precursors, and these are first converted into their amides and then dehydrated. While their functions are not yet known, some of these nitriles showed bactericidal activity; any possible homicidal properties were not investigated (Vidal, D.M. et al. Long-chain alkyl cyanides: unprecedented volatile compounds released by Pseudomonas and Micromonospora bacteria. Angew. Chem.-Int. Ed., 56, 4342-4346 (2017); DOI). Some plant species contain cyanolipids but of a very different kind.
April 5th, 2017
I have commented on the uniqueness of cardiolipin in this blog on many occasions in the past, usually in relation to its special functions in membranes. Perhaps the most distinctive feature is that it possesses a dimeric structure in essence with four acyl groups arranged in a very limited range of molecular species. In heart muscle, for example, linoleate makes up 80% of the total fatty acids, so a high proportion of the lipid exists as the tetralinoleoyl species. This composition is attained after its initial synthesis by a remodelling process, catalysed by the enzyme tafazzin, which transfers fatty acids from other phospholipids by a mechanism that does not require a coenzyme A ester as an intermediate, and it is reversible. The question has arisen as to whether the fatty acid specificity is inherent in tafazzin per se or is dependent on thermodynamic considerations. It now appears that the question has been answered definitively by two papers from the laboratory of Professor Michael Schlame (Schlame, M. and Greenberg, M.L. Biosynthesis, remodeling and turnover of mitochondrial cardiolipin. Biochim. Biophys. Acta, 1862, 3-7 (2017); DOI; Schlame, M. et al. The basis for acyl specificity in the tafazzin reaction. J. Biol. Chem., 292, 5499-5506 (2017); DOI). It seems now to be established that sufficient energy differences arise from the packing properties of the entire lipid assembly in the membrane to enable tafazzin to catalyse the remodeling of cardiolipin by combinations of forward and reverse transacylations, essentially creating an equilibrium distribution of acyl groups. The shape of tetralinoleoyl-cardiolipin is such that it fits the geometry of negatively curved monolayers particularly well so this structure is favoured.
It seems that we now have a good understanding of the nature of the complex phytoglycosphingolipids from higher plants, although much remains to be learned of their biochemistry and especially their function. However, following the recent discovery of inositol phosphorylceramide glucuronosyltransferase 1, i.e. the first enzyme in the GIPC glycosylation pathway, it has now been shown that these highly polar membrane lipids are essential for normal growth and function in Arabidopsis (Tartaglio, V. et al. Glycosylation of inositol phosphorylceramide sphingolipids is required for normal growth and reproduction in Arabidopsis. Plant J., 89, 278-290 (2017); DOI).
Older entries in this blog are archived for at least a year here..
| Author: William W. Christie | Updated: August 2nd, 2017 | Credits/disclaimer |  |